Guide/Cleaning Polluted Oxygen
Polluted Oxygen can be converted to Liquid Oxygen by cooling it down to its condensation point using an installation involving Thermo Regulators, and then reheating it. This guide describes an example system.
System described below has following characteristics:
- Uses 2650 W of energy (though it scales up and down by adding/removing Thermo Regulators)
- Can convert ~500 g/s of Polluted Oxygen (assuming 17 °C) into Oxygen
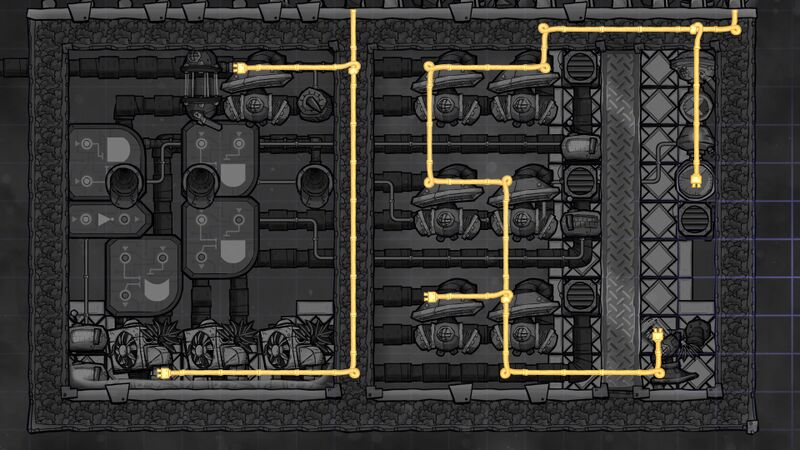
Schematics
|
|
Important details:
- The room on the left contains 50 kg/tile (350 kg total) of Petroleum. Because it has a high thermal conductivity (2 (W/m)/K) it ensures that Thermo Regulators will not overheat and nicely smooths out temperature differences. It also has a very low freezing point (-57.1 °C) which ensures that Liquid Oxygen of temperature -183 °C will not freeze it.
- The room with Hydrogen should have slightly less pressure than 20 kg (I would suggest ~18 kg) - otherwise if it's too close to 20 kg, the vents will block and the system will not perform at full efficiency.
- Use gold for most of the buildings (especially Thermo Regulator and Liquid Pump).
- I built the metal wall from Tungsten and used Diamond Tempshift Plates to ensure fast transfer of energy.
- It is important to only input Polluted Oxygen to the system - putting other gasses might break the system.
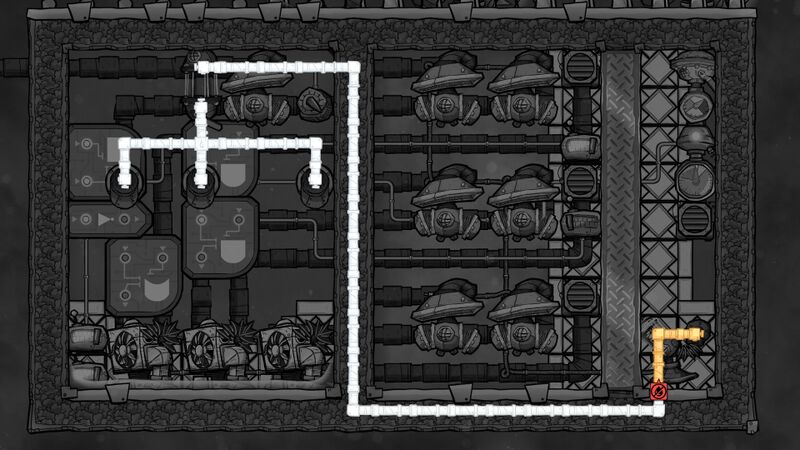
Automation
The main rule is to enable Thermo Regulators when temperature of Petroleum in the left room is lower than -45 °C (to heat it and protect against freezing) OR when temperature of Hydrogen in the middle room got higher than -200 °C (to keep it at stable temperature that can condensate Polluted Oxygen).
There is also a safety part that completely shuts off the Polluted Oxygen input when:
- Temperature of the left room is not between [-45 °C, 30 °C]
- Temperature of the middle room is lower than [-190 °C]
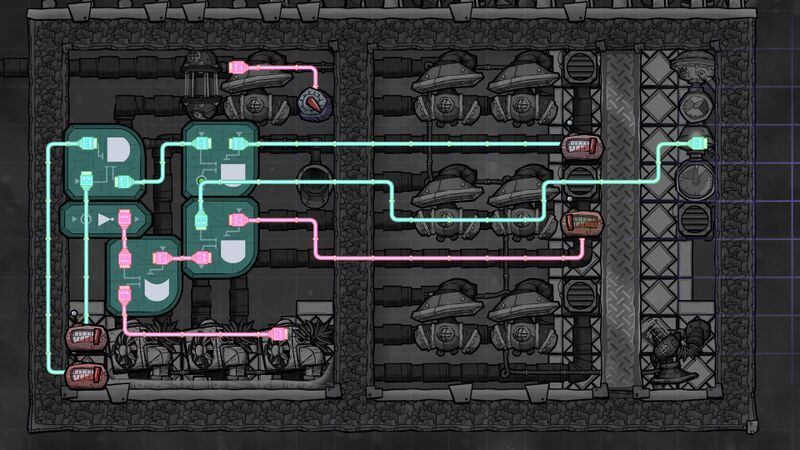
All settings, assuming that the order is from left to right, and then from up to down:
- Thermo Sensor - (Above -45 °C)
- Thermo Sensor - (Below 30 °C)
- Atmo Sensor - (Above 2000 g)
- Thermo Sensor - (Above -190 °C)
- Thermo Sensor - (Above -200 °C)
Other settings:
- Liquid Valve - (1000 g) (it is probably not needed)
- Gas Valve - (500 g) - set depending on the expected temperature of the input gas and number of Thermo Regulators
Building tips
Note: I haven't built it yet in normal mode, only debug, but this is how I plan to execute building it:
- Build the middle and right rooms.
- Fill the middle room with just hydrogen, and the room on the right should be empty
- Place thermo regulators in cold environment - I would suggest Icy Biomes or in the room with Anti Entropy Thermo-Nullifier
- Cool down the hydrogen to ~ -200 °C)
- Build a left room, pump 50 kg/tile of Petroleum (ensure that Thermo Regulators are not flooded) and close it
And you are done, pump Polluted Oxygen into the system and enjoy clean Oxygen as output.
Math
TODO: explain the math behind the system
The Thermo Regulators, via the hydrogen, are responsible for removing energy from and condensing the Polluted Oxygen. If we are given the rate in which we want to condense Polluted Oxygen (ie, the clean Oxygen rate), then the number of Thermo Regulators can be calculated.
We do this by equating the energy rate required to cool the oxygen to the energy rate required to cool the hydrogen.
Polluted Oxygen:
- Rate of Polluted Oxygen: 500 g/s
- Temperature change of Polluted Oxygen: 17-(-183) = 203 K
- Specific Heat of Polluted Oxygen: 1.01 = (W*s)/(g*K) or W = 1.01 * K * (g/s)
- Watts from Polluted Oxygen: W = 1.01 * 203 * 500 = 102515
Hydrogen:
- Temperature change of Hydrogen: 14 K
- Specific Heat of Hydrogen: 2.4 = (W*s)/(g*K) or (g/s) = W/(2.4*K)
- Rate of Hydrogen: (g/s) = 102515/(2.4*14) = 3051.04
Thermo Regulator:
- Thermo Regulator = 1000 g/s
- 3051.04 g/s * (1 Thermo Regulator)/(1000 g/s) = 3.05 Thermo Regulators